Somatostatin Receptor 2 Phosphorylation Assays
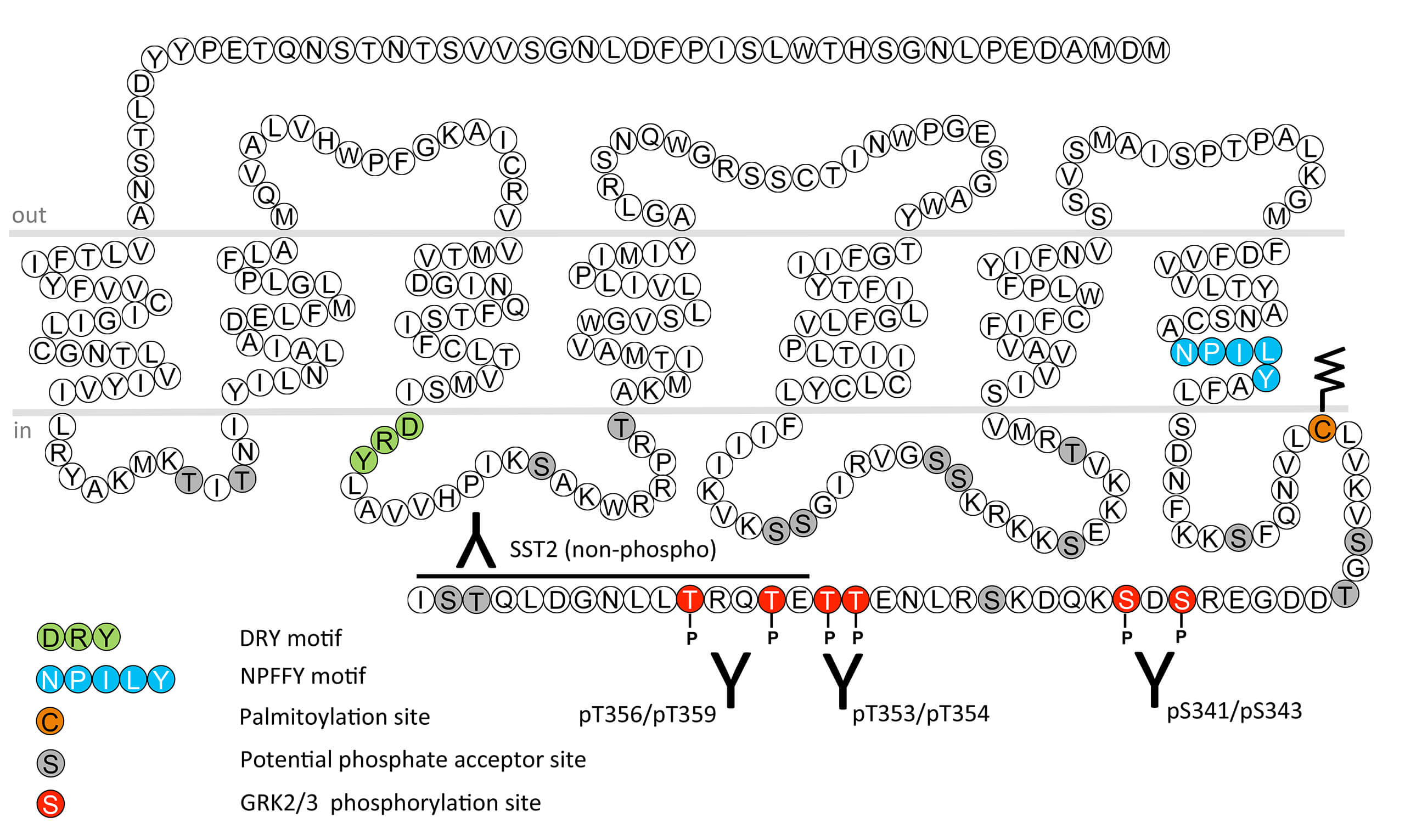
The somatostatin receptor 2 (SST2) is highly expressed in many endocrine cell types and exerts potent inhibitory actions on hormone secretion. It is the primary target for octreotide (Sandostatin) and lanreotide (Somatuline) which are the first line medical treatment for growth hormone (GH)-producing adenomas (acromegaly) and gastrointestinal neuroendocrine tumors (NET). SST2 is a prototypical GPCR for the development of peptide-based radiopharmaceuticals, which are used for imaging of human tumors as well as for peptide receptor radionuclide therapy (PRRT). SST2 desensitization, β-arrestin recruitment and internalization are regulated by phosphorylation of carboxyl-terminal serine341/serine343 (pS341/pS343-SST2), threonine353/threonine354 (pT353/pT354-SST2) and threonine356/threonine359 (pT356/pT359-SST2) residues. This nomenclature refers to the human SST2. This phosphorylation motif is highly conserved across mouse, rat and human. High-efficacy agonists such as octreotide, lanreotide, veldoreotide and SRIF induce rapid phosphorylation of all six sites which is mediated by GRK2 and GRK3. Low-efficacy agonists such as pasireotide induce primarily phosphorylation of S341/S343. S343 is also a substrate for phosphorylation by PKC. SST2 dephosphorylation is primarily mediated by protein phosphatase 1 beta (PP1β). For more information on SST2 pharmacology please refer to the IUPHAR database. For further reading refer to:
Pöll F, Lehmann D, Illing S, Ginj M, Jacobs S, Lupp A, Stumm R, Schulz S. Pasireotide and octreotide stimulate distinct patterns of sst2A somatostatinreceptor phosphorylation. Mol Endocrinol. 2010 Feb;24(2):436-46. doi: 10.1210/me.2009-0315. Epub 2010 Jan 5. PubMed PMID: 20051480; PubMed Central PMCID: PMC5428126.
Lehmann A, Kliewer A, Schütz D, Nagel F, Stumm R, Schulz S. Carboxyl-terminal multi-site phosphorylation regulates internalization and desensitization of the human sst2 somatostatin receptor. Mol Cell Endocrinol. 2014 Apr 25;387(1-2):44-51. doi: 10.1016/j.mce.2014.02.009. Epub 2014 Feb 22. PubMed PMID: 24565897.
Nagel F, Doll C, Pöll F, Kliewer A, Schröder H, Schulz S. Structural determinants of agonist-selective signaling at the sst(2A) somatostatin receptor. Mol Endocrinol. 2011 May;25(5):859-66. doi: 10.1210/me.2010-0407. Epub 2011 Feb 17. PubMed PMID: 21330405; PubMed Central PMCID: PMC5417258.
Pöll F, Doll C, Schulz S. Rapid dephosphorylation of G protein-coupled receptors by protein phosphatase 1β is required for termination of β-arrestin-dependent signaling. J Biol Chem. 2011 Sep 23;286(38):32931-6. doi: 10.1074/jbc.M111.224899. Epub 2011 Jul 27. PubMed PMID: 21795688; PubMed Central PMCID: PMC3190940.
Kliewer A, Mann A, Petrich A, Pöll F, Schulz S. A transplantable phosphorylation probe for direct assessment of G protein-coupled receptor activation. PLoS One. 2012;7(6):e39458. doi: 10.1371/journal.pone.0039458. Epub 2012 Jun 26. PubMed PMID: 22745760; PubMed Central PMCID: PMC3383726.
Lehmann A, Kliewer A, Märtens JC, Nagel F, Schulz S. Carboxyl-terminal receptor domains control the differential dephosphorylation of somatostatin receptors by protein phosphatase 1 isoforms. PLoS One. 2014 Mar 17;9(3):e91526. doi: 10.1371/journal.pone.0091526. eCollection 2014. PubMed PMID: 24637622; PubMed Central PMCID: PMC3956607.
Tulipano G, Stumm R, Pfeiffer M, Kreienkamp HJ, Höllt V, Schulz S. Differential beta-arrestin trafficking and endosomal sorting of somatostatin receptor subtypes. J Biol Chem. 2004 May 14;279(20):21374-82. Epub 2004 Mar 4. PubMed PMID: 15001578.
 pS341/pS343-SST2 Phosphorylation Assay Kit
pS341/pS343-SST2 Phosphorylation Assay Kit  pT356/pT359-SST2 Phosphorylation Assay Kit
pT356/pT359-SST2 Phosphorylation Assay Kit 

