No results were found for the filter!
NEW
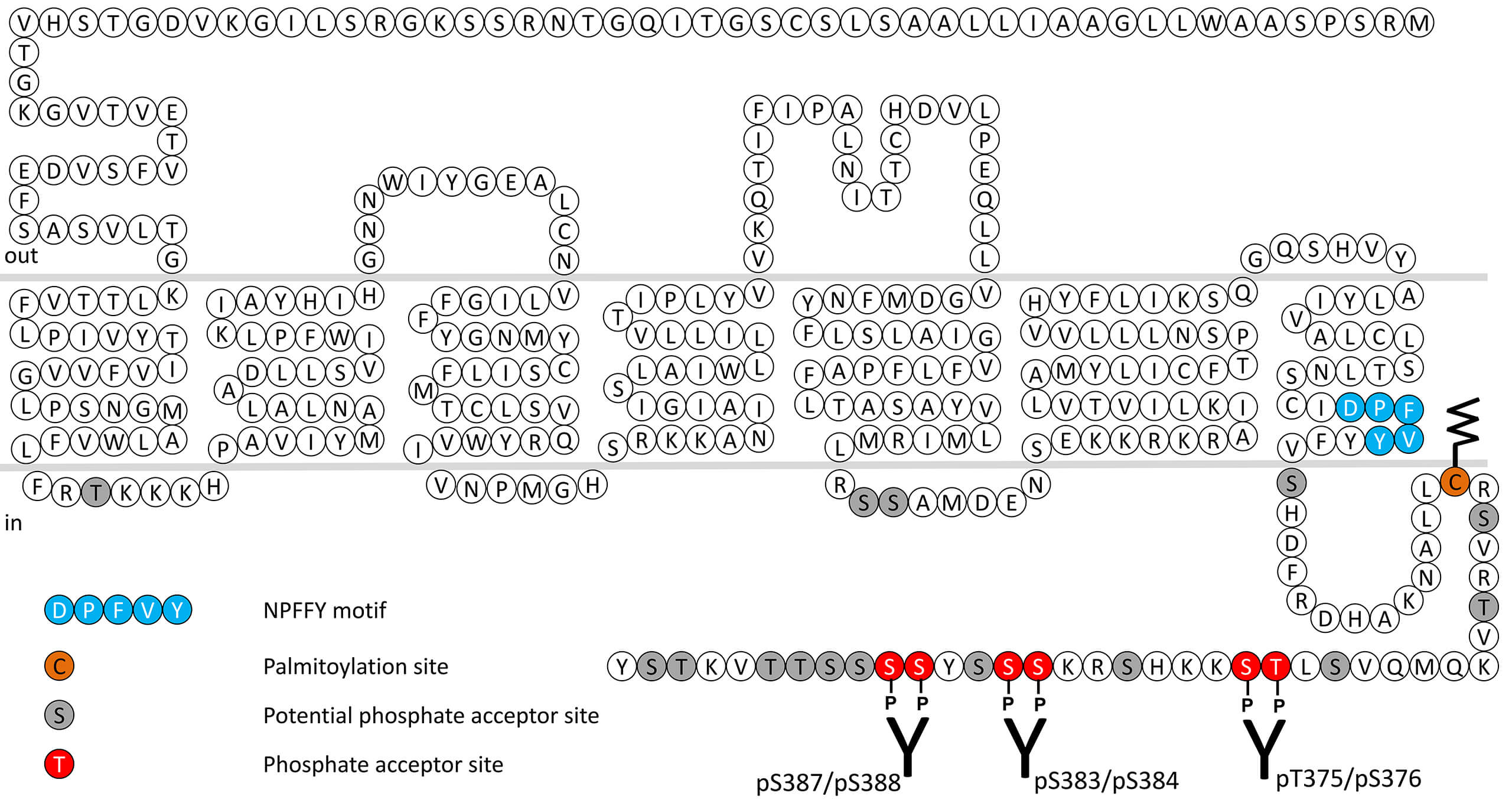
 PAR2 (IHC-grade), Proteinase-Activated Receptor...
PAR2 (IHC-grade), Proteinase-Activated Receptor... The PAR2 antibody is directed against the distal end of the carboxyl-terminal tail of mouse, rat and human PAR2. It can be used to detect total PAR2 receptors in Western blots independent of phosphorylation. The PAR2 antibody can also be...
$ 375.00 *
NEW
 pT375/pS376-PAR2 (phospho-Proteinase-Activated...
pT375/pS376-PAR2 (phospho-Proteinase-Activated... Threonine375/Serine376 (T375/S376) is major phosphorylation site of the Proteinase-Activated Receptor 2 (PAR2). The pT375/pS376-PAR2 antibody detects phosphorylation in response to agonists. T375/S376 phosphorylation is likely to be...
$ 375.00 *
NEW
 pS387/pS388-PAR2 (phospho-Proteinase-Activated...
pS387/pS388-PAR2 (phospho-Proteinase-Activated... Serine387/Serine388 (S383/S384) is major phosphorylation site of the Proteinase-Activated Receptor 2 (PAR2). The pS387/pS388-PAR2 antibody detects phosphorylation in response to agonists. S387/S388 phosphorylation is likely to be...
$ 375.00 *
NEW
 pS383/pS384-PAR2 (phospho-Proteinase-Activated...
pS383/pS384-PAR2 (phospho-Proteinase-Activated... Serine383/Serine384 (S383/S384) is major phosphorylation site of the Proteinase-Activated Receptor 2 (PAR2). The pS383/pS384-PAR2 antibody detects phosphorylation in response to agonists. S383/S384 phosphorylation is likely to be...
$ 375.00 *
NEW
 PAR2 (non-phospho), Proteinase-Activated...
PAR2 (non-phospho), Proteinase-Activated... The non-phospho-PAR2 receptor antibody is directed against the distal end of the carboxyl-terminal tail of human Proteinase-Activated Receptor 2. It can be used to detect total PAR2 receptors in Western blots independent of...
$ 375.00 *
Recently viewed