µ-Opioid Receptor Antibodies
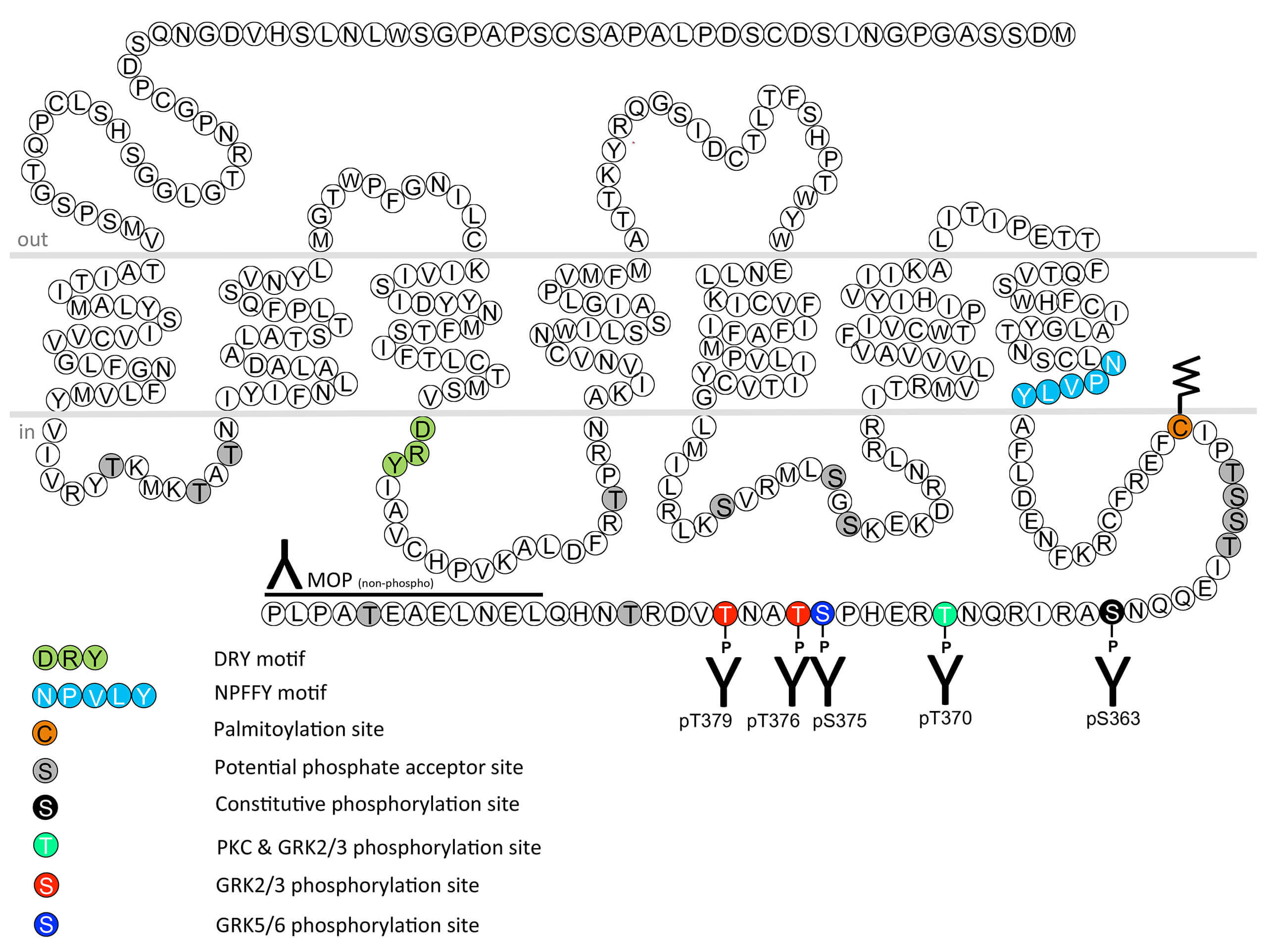
The µ-opioid receptor (mu-opioid receptor, MOP) is the primary target for many potent analgesics used in the clinic including morphine, oxycodone, hydromorphone, fentanyl, sufentanil and methadone. The utility of µ-analgesics is limited by sever side effects such as respiratory depression, constipation, dependence and addiction. DAMGO ([D-Ala2,N-MePhe4, Gly-ol]-enkephalin) is an enkephalin derivative that is used as selective µ-agonist for in vitro studies. MOP desensitization, trafficking and tolerance are regulated by phosphorylation of carboxyl-terminal threonine370 (pT370-MOP), serine375 (pS375-MOP), threonine376 (pT376-MOP) and threonine379 (pT379-MOP) residues. This nomenclature refers to the mouse MOP. The human MOP sequence is highly conserved at this phosphorylation motif, however, individual sites correspond to T372, S377, T378 and T381 due to two additonal amino acid residues at the amino terminus in the human sequence. S375 is the primary site in a hierarchical phosphorylation cascade. High-efficacy agonists such as DAMGO, fentanyl, sufantanil and methadone induce phosphorylation of all four sites which is mediated by GRK2 and GRK3. Low-efficacy agonists such as morphine, oxycodone and hydromorphone induce primarily phosphorylation of S375 that is mediated in part by GRK5. T370 is subject to phosphorylation by PKC. Serine363 (pS363-MOP) is constitutively phosphorylated by PKC. Rapid MOP dephosphoryation is primarily mediated by protein phosphatase 1 gamma (PP1γ). The development of G protein-biased µ-agonists such as TRV130, PZM21 or SR-17018 has been proposed as a strategy for safer analgesics with fewer side effects. Recent work shows that phosphosite-specific antibodies provide more precise information about ligand bias then conventional ß-arrestin recruitment assays. For more information on µ-opioid receptor pharmacology please refer to the IUPHAR database. For further reading refer to:
Fritzwanker S, Nagel F, Kliewer A, Schulz S. In situ Visualization of therapeutic drug effects using phosphosite-specific GPCR antibodies. bioRxiv 2022.06.14.496067; doi: https://doi.org/10.1101/2022.06.14.496067
Kliewer A, Schmiedel F, Sianati S, Bailey A, Bateman JT, Levitt ES, Williams JT, Christie MJ, Schulz S. Phosphorylation-deficient G-protein-biased μ-opioid receptors improve analgesia and diminish tolerance but worsen opioid side effects. Nat Commun. 2019 Jan 21;10(1):367. doi: 10.1038/s41467-018-08162-1. PubMed PMID: 30664663; PubMed Central PMCID: PMC6341117.
Miess E, Gondin AB, Yousuf A, Steinborn R, Mösslein N, Yang Y, Göldner M, Ruland JG, Bünemann M, Krasel C, Christie MJ, Halls ML, Schulz S, Canals M. Multisite phosphorylation is required for sustained interaction with GRKs and arrestins during rapid μ-opioid receptor desensitization. Sci Signal. 2018 Jul 17;11(539). pii: eaas9609. doi: 10.1126/scisignal.aas9609. PubMed PMID: 30018083.
Yousuf A, Miess E, Sianati S, Du YP, Schulz S, Christie MJ. Role of Phosphorylation Sites in Desensitization of µ-Opioid Receptor. Mol Pharmacol. 2015 Oct;88(4):825-35. doi: 10.1124/mol.115.098244. Epub 2015 May 12. PubMed PMID: 25969388.
Glück L, Loktev A, Moulédous L, Mollereau C, Law PY, Schulz S. Loss of morphine reward and dependence in mice lacking G protein-coupled receptor kinase 5. Biol Psychiatry. 2014 Nov 15;76(10):767-74. doi: 10.1016/j.biopsych.2014.01.021. Epub 2014 Feb 3. PubMed PMID: 24629717; PubMed Central PMCID: PMC4119866.
Illing S, Mann A, Schulz S. Heterologous regulation of agonist-independent μ-opioid receptor phosphorylation by protein kinase C. Br J Pharmacol. 2014 Mar;171(5):1330-40. doi: 10.1111/bph.12546. PubMed PMID: 24308893; PubMed Central PMCID: PMC3952808.
Just S, Illing S, Trester-Zedlitz M, Lau EK, Kotowski SJ, Miess E, Mann A, Doll C, Trinidad JC, Burlingame AL, von Zastrow M, Schulz S. Differentiation of opioid drug effects by hierarchical multi-site phosphorylation. Mol Pharmacol. 2013 Mar;83(3):633-9. doi: 10.1124/mol.112.082875. Epub 2012 Dec 13. PubMed PMID: 23239825; PubMed Central PMCID: PMC3583494.
Doll C, Pöll F, Peuker K, Loktev A, Glück L, Schulz S. Deciphering µ-opioid receptor phosphorylation and dephosphorylation in HEK293 cells. Br J Pharmacol. 2012 Nov;167(6):1259-70. doi: 10.1111/j.1476-5381.2012.02080.x. PubMed PMID: 22725608; PubMed Central PMCID: PMC3504992.
Grecksch G, Just S, Pierstorff C, Imhof AK, Glück L, Doll C, Lupp A, Becker A, Koch T, Stumm R, Höllt V, Schulz S. Analgesic tolerance to high-efficacy agonists but not to morphine is diminished in phosphorylation-deficient S375A μ-opioid receptor knock-in mice. J Neurosci. 2011 Sep 28;31(39):13890-6. doi: 10.1523/JNEUROSCI.2304-11.2011. PubMed PMID: 21957251; PubMed Central PMCID: PMC6633166.
Doll C, Konietzko J, Pöll F, Koch T, Höllt V, Schulz S. Agonist-selective patterns of µ-opioid receptor phosphorylation revealed by phosphosite-specific antibodies. Br J Pharmacol. 2011 Sep;164(2):298-307. doi: 10.1111/j.1476-5381.2011.01382.x. PubMed PMID: 21449911; PubMed Central PMCID: PMC3174411.
 pS375-MOP (IHC-grade phospho-µ-Opioid Receptor...
pS375-MOP (IHC-grade phospho-µ-Opioid Receptor...  pT376-MOP (IHC-grade phospho-µ-Opioid Receptor...
pT376-MOP (IHC-grade phospho-µ-Opioid Receptor...  pT379-MOP (IHC-grade phospho-µ-Opioid Receptor...
pT379-MOP (IHC-grade phospho-µ-Opioid Receptor... 

